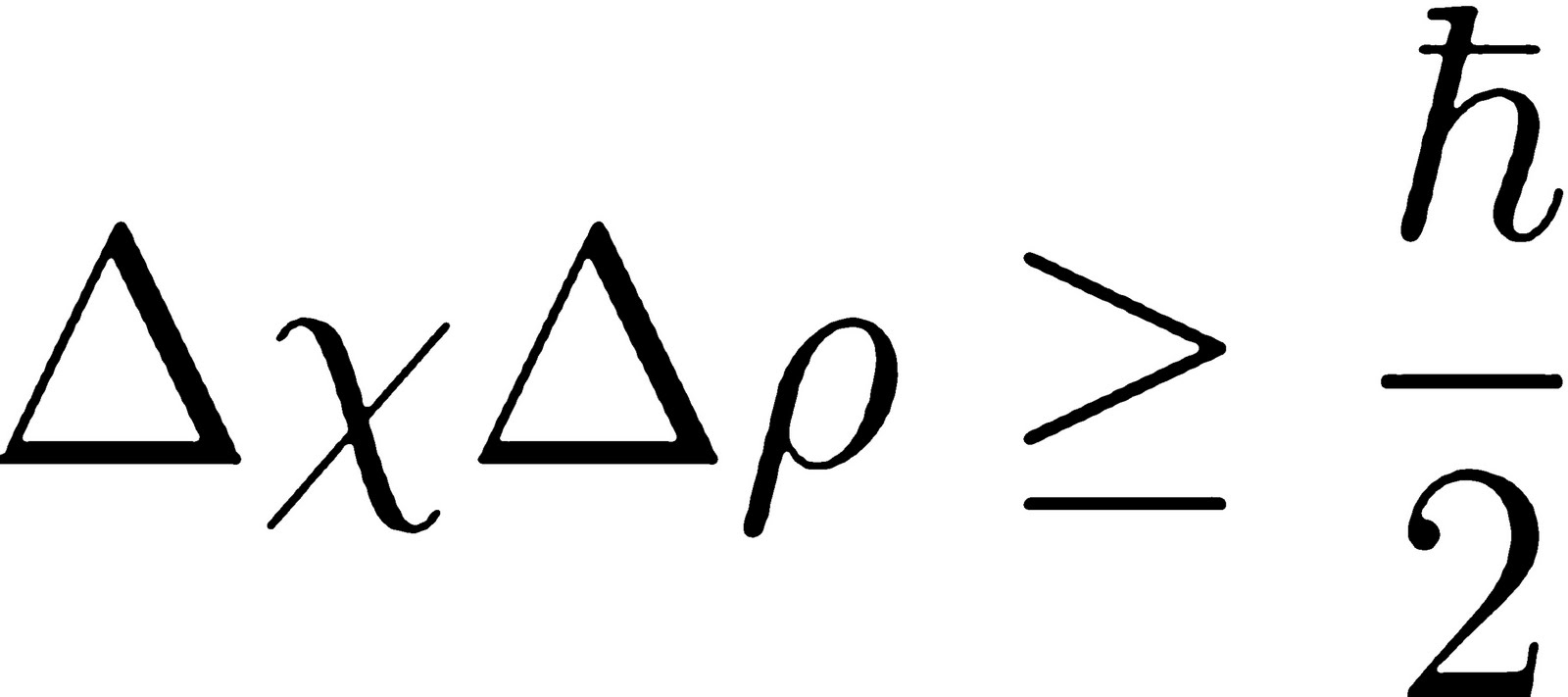
Though not imparting as much disruption to the electron's momentum, a longer wavelength of light wouldn't allow as precise a measurement. The smallest wavelength of light, called gamma-ray light, can make the most precise measurements, but it also carries the most energy, so an impacting gamma-ray photon will deliver a stronger kick to the electron, thereby disturbing its momentum the most. 
The wavelength of the light determines how precisely the measurement can be made. When a photon, or particle of light, hits the electron, it will bounce back and record its position, yet in the process of doing so, it has given the electron a kick, thereby changing its speed. Imagine shining light at a moving electron. Heisenberg originally explained the limitation using a thought experiment. 
Or you might choose to determine an electron's momentum fairly precisely, but then you will have only a vague idea of its location. Its logic is perplexing to the human mind, which is acclimated to the macroscopic world, where measurements are only limited by the quality of our instruments.īut in the microscopic world, there truly is a limit to how much information we can ever glean about an object.įor example, if you make a measurement to find out exactly where an electron is, you will only be able to get a hazy idea of how fast it's moving. The uncertainty principle only applies in the quantum mechanical realm of the very small, on scales of subatomic particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
